Background: Oral ABL-targeted kinase inhibitors (TKIs) have transformed treatment of BCR::ABL+ (Philadelphia chromosome-positive, Ph+) acute lymphoblastic leukemia (ALL). Induction (IND) w/ corticosteroids (CS) and dasatinib (DAS) alone results in morphologic complete response (mCR) rates approaching 100% but low rates of measurable residual disease (MRD) negativity; addition of intensive chemotherapy to TKIs adds risks of myelosuppression. DAS + the CD3/CD19 bispecific T-cell engager blinatumomab (BLIN) is effective consolidation (CONSOL) for Ph+ ALL, though ABL kinase mutations conferring resistance can arise early in therapy (Foà, NEJM, 2020). We designed a phase II study of BLIN as part of a chemotherapy sparing strategy in pts w/ Ph+ ALL (BLISSPHALL), introducing BLIN as early as 6 weeks into treatment for pts in mCR, w/ aim of expediting MRD clearance and suppressing resistant clones early in disease course, and including a maintenance (MAINT) phase of BLIN + TKI for pts in molecular response.
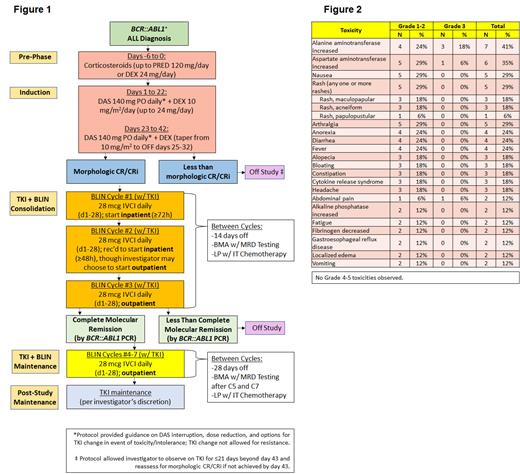
Methods: We conducted a multicenter trial of TKI + CS IND and TKI + BLIN CONSOL + MAINT in adults (≥18 yrs) w/ newly-diagnosed (ND) Ph+ ALL (NCT04329325). Study schema is depicted in Figure 1. Pts received CS pre-phase w/ prednisone (PRED) or dexamethasone (DEX) followed by modified GIMEMA LAL1205 IND (DEX + DAS 140 mg/d w/ DAS dose adjustments or TKI change per protocol) w/ intrathecal methotrexate (IT MTX) d22, 43 and bone marrow (BM) MRD assessments including FACS and BCR::ABL1 PCR. Pts in mCR on d43 could proceed to CONSOL w/ 3 cycles (C) of TKI + BLIN 28 mcg/d IVCI, d1-28, w/ 14d off BLIN between cycles and BM MRD assessment/IT MTX after each cycle. TKI was given continuously including between BLIN cycles. Pts in protocol-defined complete molecular response (CMR; MRD negative by FACS and no detectable BCR::ABL1 transcripts) after CONSOL could proceed to MAINT (C4-7 BLIN + TKI, 28d off between cycles). The primary objective is to determine the proportion of evaluable pts achieving CMR by the end of CONSOL (≤ 3C BLIN + TKI). Secondary objectives include safety/toxicity of BLIN + DAS, duration of CMR, incidence of relapse, event-free/overall survival. Toxicities were defined by CTCAE v5.0.
Results: 17 pts enrolled (12 women, 5 men) w/ median age 50 yrs (range 22-87); BCR::ABL1 transcript type p190 (n=15) or p210 (n=2). Median follow-up 11.7 months (range 3-24). All began DAS 140 mg/d as initial TKI; 4 changed TKI for DAS intolerance per protocol (bosutinib, n=2; ponatinib [PON], n=2) and remained on study; TKI was changed to PON in 1 other pt to optimize BCR::ABL1 transcript suppression pre-allogeneic transplant (alloHCT) and that pt was withdrawn from study. Two had interruption or delay of BLIN due to toxicity (grade [G] 3 transaminase elevation, n=1; G2 cytokine release syndrome, n=1); BLIN was resumed in both. Figure 2 lists nonhematologic adverse effects (AEs) seen in ≥2 pts at least possibly related to TKI or BLIN while on study. One pt developed G3 pancreatitis after DAS was changed to PON; TKI was changed back to DAS. There were no G4-5 AEs.
All pts (100%) achieved mCR during IND (median d22). Ten achieved CMR to date (5 during IND, 5 during CONSOL) and 6 did not achieve CMR; determination pending in 1 pt. By FACS, 16/17 achieved BM MRD negativity; 13/14 evaluable pts had no evidence of malignant clonal IgH rearrangement (IgHR) by next generation sequencing (NGS). Notably, 3 pts w/ persistent low-level BCR::ABL1 transcripts had no evidence of malignant clonal IgHR by NGS but were withdrawn post-CONSOL as protocol required BCR::ABL1 PCR negativity to proceed to MAINT. Two pts relapsed, both after achieving CMR (one declined CONSOL and self-discontinued TKI; one had extramedullary (EM) relapse w/ ABL T315I mutation). Four pts underwent alloHCT in CR1 (rising MRD levels on study leading to change in therapy, n=1; persistent BCR::ABL1 PCR positivity, n=2; IKZF1 plus phenotype, n=1); one underwent alloHCT in CR2 following EM relapse.
Conclusions: DAS + CS IND w/ addition of BLIN to TKI in mCR leads to high rates of deep molecular response w/ low risk of early relapse and low rates of severe regimen-related toxicity in pts w/ ND Ph+ ALL. Use of PON (vs DAS) as initial TKI w/ BLIN may further suppress resistant clones. Further f/u is needed to confirm durability of CMR w/ BLIN + TKI CONSOL ± MAINT in absence of alloHCT and whether pts w/ persistent BCR::ABL1 PCR positivity and no malignant clonal IgHR by NGS exhibit similar outcomes to those in CMR.
OffLabel Disclosure:
Geyer:Sanofi: Consultancy, Research Funding; Novartis: Consultancy; Actinium Pharmaceuticals, Inc: Research Funding; Amgen: Research Funding. Mascarenhas:Bristol Myers Squibb, Celgene, CTI BioPharma, Geron, Incyte Corporation, Janssen, Kartos Therapeutics, Merck, Novartis, PharmaEssentia, Roche; Participated in consulting or advisory committees - AbbVie, Bristol Myers Squibb, Celgene, Constellation Pharmac: Research Funding; Incyte, Novartis, Roche, Geron, GSK, Celgene/BMS, Kartos, AbbVie, Karyopharm, PharmaEssentia, Galecto, Imago, Sierra Oncology, Pfizer, MorphoSys, CTI Bio: Consultancy; Bristol Myers Squibb, Celgene, Constellation Pharmaceuticals/MorphoSys, CTI BioPharma, Galecto, Geron, GSK, Incyte Corporation, Karyopharm Therapeutics, Novartis, PharmaEssentia, Prelude Therapeutics, Pfizer, Merck, Roche, AbbVie, Kartos: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie, Bristol Myers Squibb, Celgene, CTI BioPharma, Geron, Incyte Corporation, Novartis, Janssen, Kartos Therapeutics, Merck, PharmaEssentia, Roche: Research Funding; GSK: Honoraria; AbbVie, CTI BioPharma Corp, a Sobi company, Geron, GlaxoSmithKline, Imago, Incyte, Kartos, Kayropharm, MorphoSys, Novartis, Pfizer, PharmaEssentia, Sierra: Consultancy. Thompson:AstraZeneca: Research Funding; Genentech: Research Funding; Nurix Therapeutics: Research Funding; Abbvie: Research Funding; Genmab: Research Funding; Brazilian Association of Hematology, Hemotherapy and Cellular Therapy (ABHH): Honoraria; Massachusetts Medical Society: Honoraria; MJH Life Sciences: Honoraria; Intellisphere LLC: Honoraria; Curio Science: Honoraria; Dava Oncology: Other: Travel ; Loxo Oncology at Lilly: Consultancy; AstraZeneca: Consultancy; Janssen: Consultancy; VJHemOnc: Honoraria; Beigene: Research Funding. Park:BeiGene: Consultancy; Servier: Consultancy, Research Funding; Sobi: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Allogene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy; Genentech, Inc.: Research Funding; Incyte: Research Funding; GC Cell: Membership on an entity's Board of Directors or advisory committees; Fate Therapeutics: Research Funding; Autolus Therapeutics: Research Funding; Kite: Consultancy; Minerva Bio: Consultancy; Be Biopharma: Consultancy; Intella: Consultancy; Amgen: Consultancy; Bright Pharmacetuicals: Consultancy; Curocell: Consultancy; Artiva Biotherapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Affyimmune: Consultancy.
Dasatinib (not FDA approved for newly-diagnosed acute lymphoblastic leukemia); blinatumomab (not FDA approved as consolidation for acute lymphoblastic leukemia without minimal residual disease, or in combination with tyrosine kinase inhibitor)